 This article was written for you by Samantha, one of the tutors with TestPrep Academy. Solution: As we move down a group, the atomic radius grows due to the filling of an extra shell with electrons. Looking to get ready for the ACT? We can help with ACT Prep It is important to note that elements in the same period all have the same number of electrons shells, so electron shielding will not be a factor.Ītomic size increases as you go down a column because of the addition of another electron shell and electron shielding.Ītomic size decreases as you go right across a row because of increased protons. Thus, the electrons are held more tightly towards the nucleus, decreasing the radius. 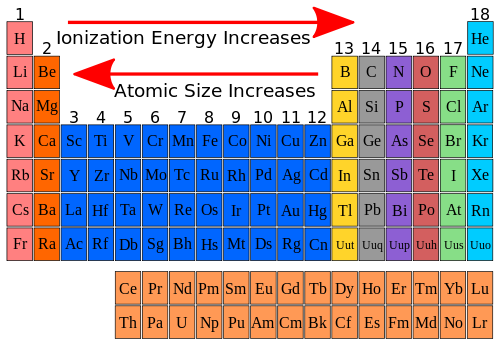
The increase in positive charge increases the attraction between the nucleus and the electrons of the atom. As the number of protons increase, the nucleus of the atom becomes more positively charged. This is because the number of protons increase moving to the right of the row. When moving left to right across a period, the atomic size decreases. Going Left and Right Across Periods (Rows) As a result, the electrons are not held as tightly towards the nucleus. 
In addition, the electron shells in between the valence shell and nucleus present electron shielding that also minimizes the attraction. For example, the covalent radius of carbon is 0.77 and the covalent radius of bromine is 1.14. Atomic size is difficult to measure because it has. A remarkably good approximation of single bond distances in compounds can be obtained by taking the sum of covalent radii. Atomic size is the distance from the nucleus to the valence shell where the valence electrons are located. Also, the new orbital is further away from the nucleus, meaning the attraction between the positively charged nucleus and the valence electrons decrease. For instance, the I - I distance in I 2 is 2.66 so the covalent radius of I is 1.33. This is because as you go down the period table, new valence shells are added and thus, increasing the radius. When moving down a group, the atomic size increases. 3.9K 268K views 5 years ago New AP & General Chemistry Video Playlist This chemistry video tutorial provides a basic introduction into atomic radius which is one of the four main periodic table. The atomic size, or atomic radius, is the distance between the nucleus of an atom to the outermost electron orbital, where the valence electrons are. These patterns are called periodic trends. The radius of an atom can increase or decrease when it becomes an ion, and this is known as the atom’s ionic radius. The atomic radius of a neutral atom equals its diameter. If an atom receives or loses an electron and becomes positively or negatively charged, it forms an ion. The tendencies of these patterns increase or decrease as you move along the columns and rows of the table. The atomic and ionic radii of atoms differ. 
Specific patterns of certain elemental characteristics are present in the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
